| Alternative names | N/A |
| Known ligands | HVEM |
| Origin | Homo sapiens |
| Accession number | Q7Z6A9 |
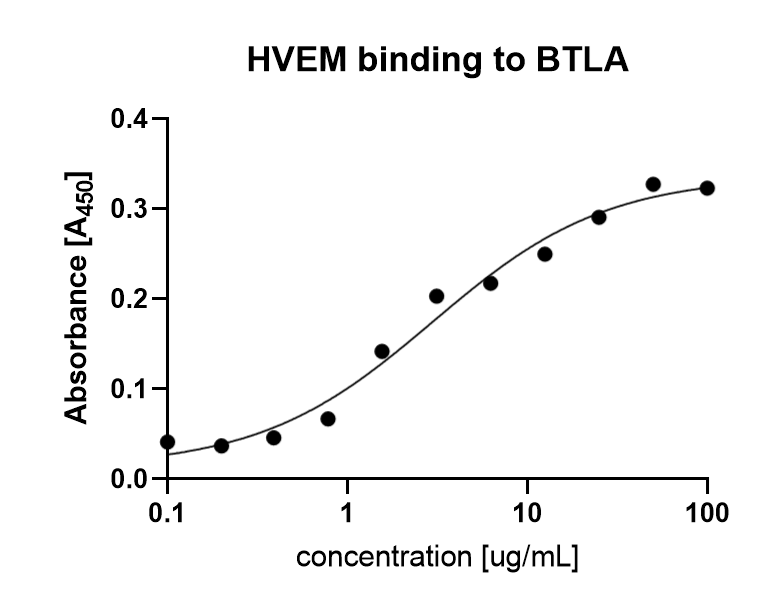
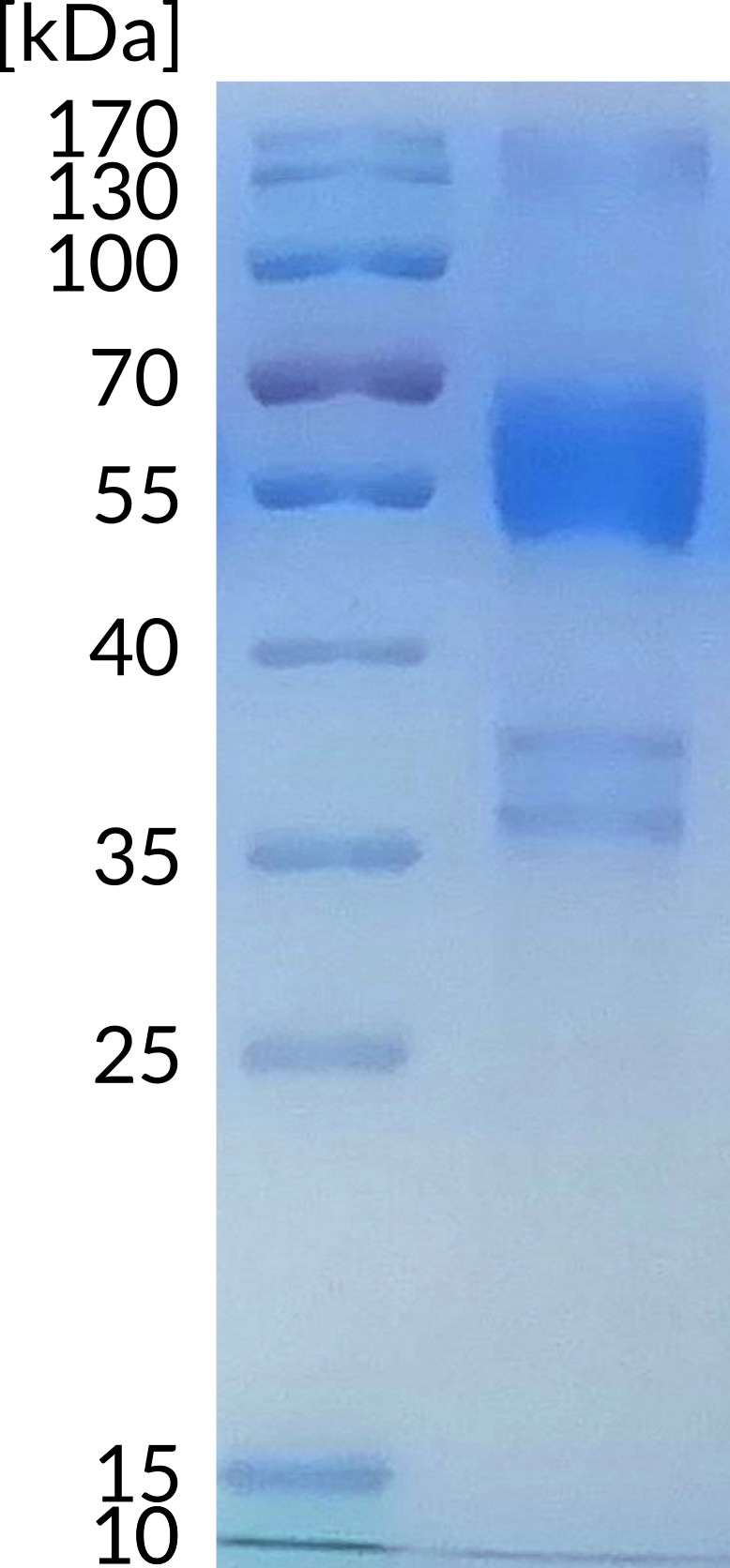
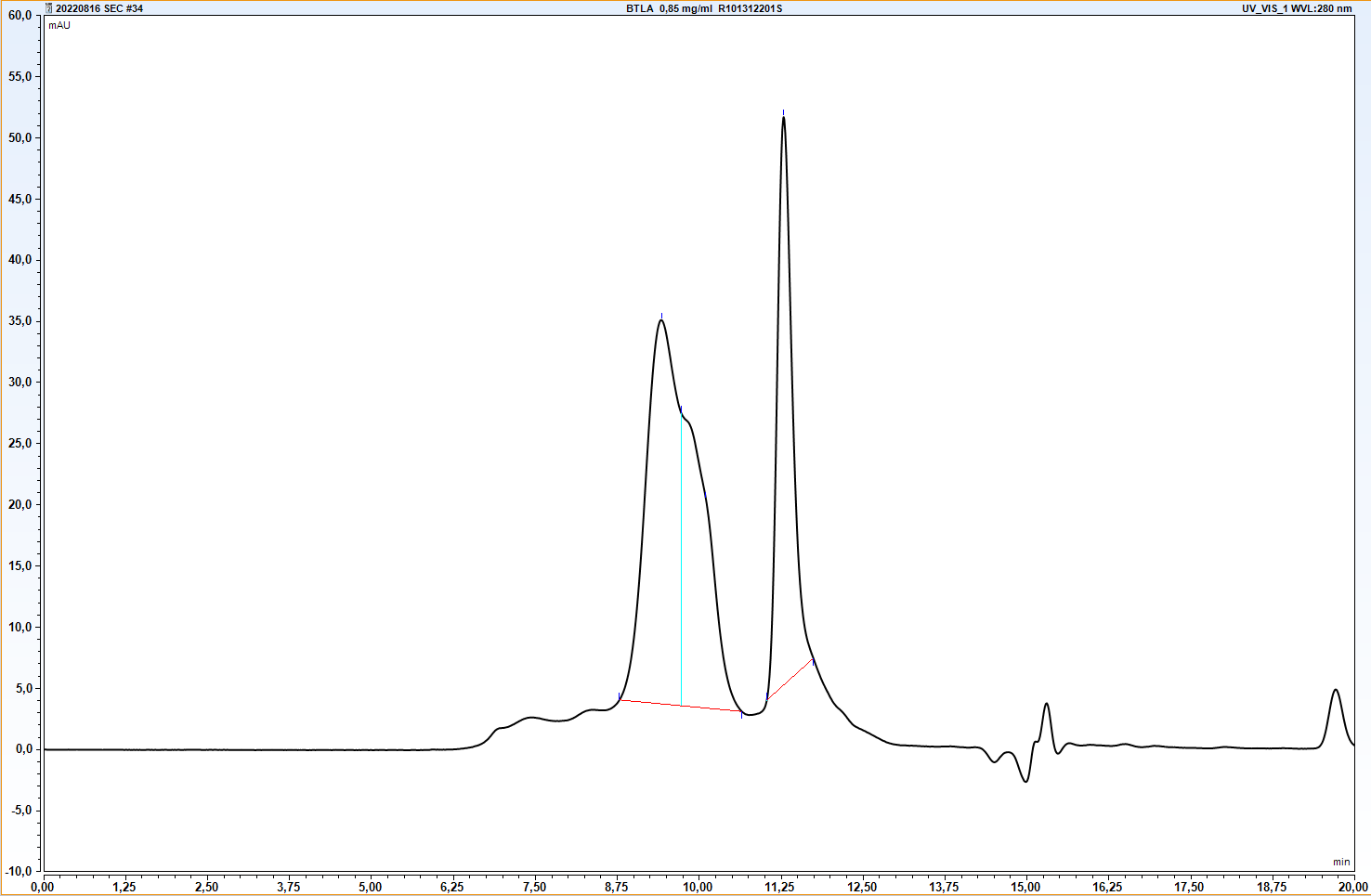

B- and T-lymphocyte attenuator (BTLA) is a 259 aminoacid protein, that exhibits compact IgG domain structure, comprised of two flat β-sheets (1). BTLA binds to HVEM. This interaction regulates immune response negatively or positively, depending on wheter the interaction is cis (both receptors are present on the surface of the same cell) or trans (interacting receptors are present on different cells). Cis interaction between BTLA and HVEM inhibits trans activation from adjacent cells, therefore maintaining T cells in the naive state (2). BTLA is found primarily in thymus, although lower levels are also observed in bone marrow. It is constitutively expressed on peripheral naive B cells, whereas lower intensity of expression is observed on the surface of naive T cells, macrophages, DC, NKT and NK cells. It can be upregulated within hours on activated human Th-1 and Th-2 polarized CD4 T cells (3).
- Compaan, D. M., Gonzalez, L. C., Tom, I., Loyet, K. M., Eaton, D., & Hymowitz, S. G. (2005). Attenuating lymphocyte activity: the crystal structure of the BTLA-HVEM complex. Journal of Biological Chemistry, 280(47), 39553-39561.
- Cheung, T. C., Oborne, L. M., Steinberg, M. W., Macauley, M. G., Fukuyama, S., Sanjo, H., … & Ware, C. F. (2009). T cell intrinsic heterodimeric complexes between HVEM and BTLA determine receptivity to the surrounding microenvironment. The Journal of Immunology, 183(11), 7286-7296.
- Del Rio, M. L., Lucas, C. L., Buhler, L., Rayat, G., & Rodriguez‐Barbosa, J. I. (2010). HVEM/LIGHT/BTLA/CD160 cosignaling pathways as targets for immune regulation. Journal of leukocyte biology, 87(2), 223-235.
